
Mårtensson, "Core-Level Binding Energies in Metals," J. Summary: Atomic Structure Atoms and Atomic Orbitals Electron Configuration and Valence Electrons Periodic Trends Terms Topics Electron Configuration and Valence Electrons Electron Configuration The electrons in an atom fill up its atomic orbitals according to the Aufbau Principle 'Aufbau,' in German, means 'building up. Lide, (Ed.) in Chemical Rubber Company handbook of chemistry and physics, CRC Press, Boca Raton, Florida, USA, 81st edition, 2000. Ley, Eds., Photoemission in Solids I: General Principles (Springer-Verlag, Berlin) with additional corrections, 1978. Cu has a unique fully-filled 3d configuration in its ground state and so has unique. Burr, "Reevaluation of X-Ray Atomic Energy Levels," Rev. Copper Table of Contents Chemical Properties Of Copper What is Copper Density of Copper Important Uses Of Copper Certain Facts About Copper Frequently Asked Questions FAQs Chemical Properties Of Copper What is Copper Copper is a chemical element with atomic number 29 and symbol Cu. The electron configuration of copper is : 1s2 2s2 2p6 3s2 3p6 4s1 3d10. They are tabulated elsewhere on the WWW (reference 4) and in paper form (reference 5). The data are adapted from references 1-3. When we write the configuration well put all 26 electrons in orbitals around the nucleus of the Iron atom. Once we have the configuration for Fe, the ions are simple. I am grateful to Gwyn Williams (Jefferson Laboratory, Virginia, USA) who provided the electron binding energy data. We first need to find the number of electrons for the Cu atom (there are 29 electrons) using the Periodic Table. In order to write the Iron electron configuration we first need to know the number of electrons for the Fe atom (there are 26 electrons). 
The binding energies are quoted relative to the vacuum level for rare gases and H 2, N 2, O 2, F 2, and Cl 2 molecules relative to the Fermi level for metals and relative to the top of the valence band for semiconductors. Electronic configuration of Copper: The atomic number of Copper ( Cu) 29 Therefore, the expected electronic configuration is Ar 3 d 9 4 s 2. All values of electron binding energies are given in eV. Exceptions Chromium and copper have the following electron configurations, which are different to what you may expect: Cr is Ar 3d5 4s1 not Ar 3d4 4s Cu. Electronic configuration: Electronic configuration is the distribution of the electrons in orbitals of atoms using some basic principles like the Pauli exclusion principle and the Aufbau principle. 1967, 47, 1300.Įlectron binding energies Electron binding energies for copper. The electron configuration of copper ion shows that copper ion(Cu +) has three shells and the last shell has eighteen electrons(3s 2 3p 6 3d 10). Then the relative energies of 4s and 3d switch. 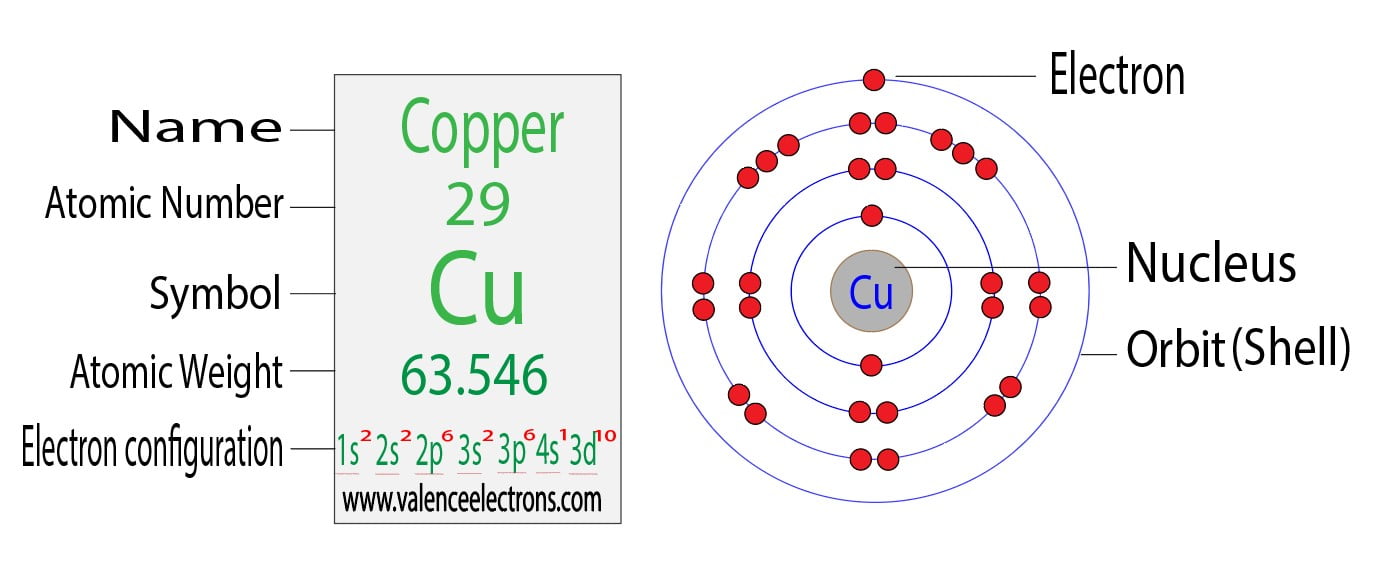
Cu e Cu + Here, the electron configuration of copper ion(Cu +) is 1s 2 2s 2 2p 6 3s 2 3p 6 3d 10. ( 91 votes) Ernest Zinck 8 years ago 4s is higher in energy than 3d until you get to Ca. Atomic no.These effective nuclear charges, Z eff, are adapted from the following references: The copper atom donates an electron in the 4s orbital to form a copper ion(Cu +). The Shorthand electron configuration (or Noble gas configuration) as well as Full electron configuration is also mentioned in the table. 
Electron configuration chart of all Elements is mentioned in the table below. Copper, silver, and gold are in group 11 of the periodic table these three metals have one s-orbital electron on top of a filled d- electron shell and are characterized by high ductility, and electrical and thermal conductivity.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
